Published on 27 March 2024
Controlled microfluidic creation for spatiotemporal pH control and particle synthesis in double emulsions
The present work performed in the department of chemical and biological engineering at the University of Sheffield, describes how the researchers used pressure-driven droplet microfluidics in developing a system of enzyme-encapsulated double emulsion droplets to obtain a spatiotemporal pH control.
The research summary written by Rana Maheen is based on the peer-reviewed article “A microfluidic double emulsion platform for spatiotemporal control of pH and particle synthesis” by Rana Maheen, Ahmad Raheel, and Annette Taylor. Their work was recently published in Lab on a chip, 2023, 23, 4504-4513.



Abstract
Controlling pH dynamics in microreactors, especially in emulsion droplets, is crucial for various applications such as biomineralisation and microparticle synthesis. In this study, Rana et al. utilize an enzyme reaction (urea-urease) in a customizable water-oil-water double emulsion (W/O/W) platform to precisely engineer the pH-time profile in droplets. A base pulse (ammonia) triggered by adding urea led to a pH increase to 8 within the droplets, with a time delay of the order of minutes. Manipulation of the pH-time profile was achieved by tuning the diffusion time scale of urea and ammonia through the oil layer. This resulted in a precise steady-state pH not observed in bulk solutions. This work demonstrates the potential for precise pH control in microreactors and opens avenues for customized synthesis of pH-sensitive materials.
Introduction
Controlling pH in microreactors, such as emulsion droplets, is crucial for various applications such as material synthesis and biomineralisation. Conventional methods often rely on passive diffusion or enzymatic reactions in vesicles. Here we present a new approach using water-oil-water double emulsions (W/O/W) to precisely modulate pH in microdroplets.
By encapsulating the urease enzyme in the aqueous core of the double emulsions, we were able to generate localised ammonia pulses that allow for a tunable steady-state pH in the droplets. Microfluidic techniques facilitated the preparation of uniform double emulsions and ensured high encapsulation efficiency and reproducibility.
Coupling urease-mediated pH modulation with the precipitation of calcium phosphate in the droplets demonstrated the versatility of our platform. By fine-tuning the thickness of the oil shell, we were able to selectively obtain the desired particles and avoid undesired results. Our microfluidic double emulsion platform provides spatio-temporal control over pH and facilitates material synthesis under mild conditions.
Aims
- Develop and fine-tune a platform with synthetic bottom-up chemistry that enables us to systematically and thoroughly investigate the effects of entrapment on a feedback-driven enzymatic reaction (urea-urease).
- Benefit from pressure driven droplet microfluidics to develop a system of enzyme-encapsulated (urea-urease) double emulsion (W/O/W) droplets to obtain a localized pH pulse with a controllable induction time to program material properties such as the formation of inorganic particles.
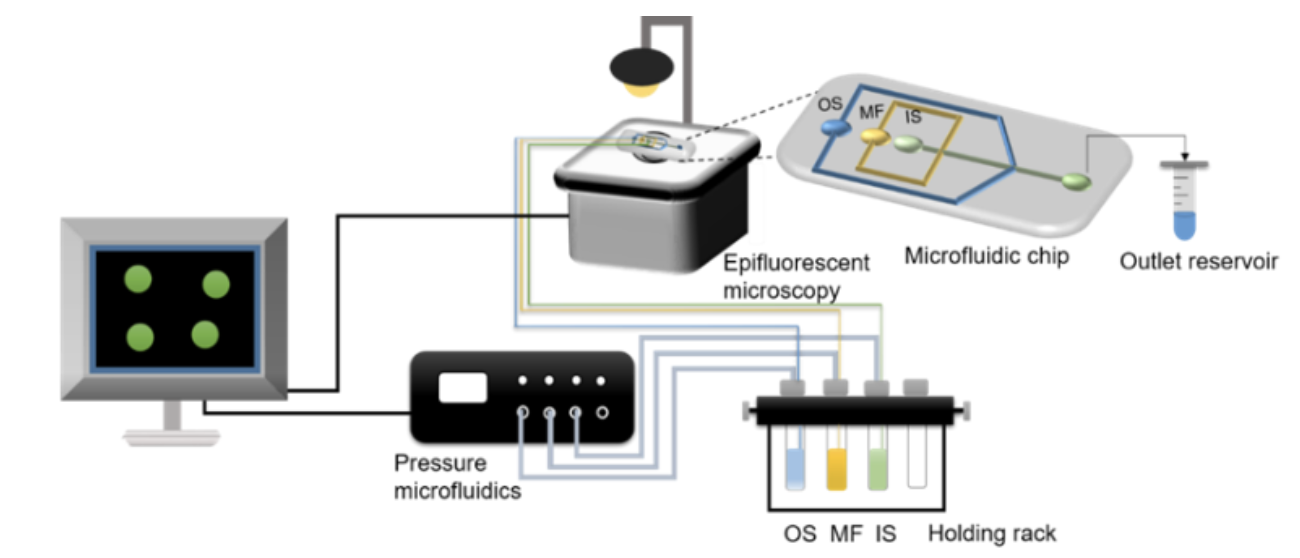
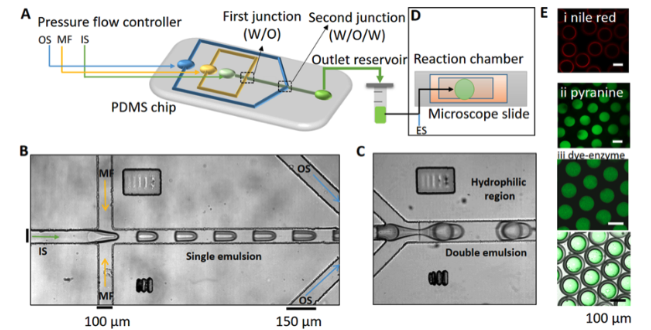
Experiment Setup | On-chip generation of W/O/W double emulsions
We used a flow-focusing device with two droplet formation junctions to synthesize the double emulsion (Fig. 1 and 2A). The polydimethylsiloxane (PDMS) micro-channel was fabricated using conventional soft lithography [1, 2]. The first junction of the channel has a hydrophobic surface to allow the formation of W/O (water in oil) droplets while the second junction is hydrophilic (PVA coated) to preferentially make O/W (oil in water) droplets, thus forming W/O/W double emulsions at the end of the microfluidic device [3, 4]. A pressure-driven pump (OB1 MK3+, Elveflow, Paris, France) was used to transport both the aqueous inner and outer solutions (IS and OS) and the lipid-oil middle fluid (MF) at a persistent flow, and a 2 mL Eppendorf tube was used as a collection vial for the double emulsions. The synthesis of the microfluidic double emulsions was visualized on a microscope using the built-in Elveflow Software Interface (ESI).
Reaction observation
For the reaction, the double emulsions were mixed with an equal volume of an external solution (ES) containing acetic acid (1 or 2 mM), glucose (0.2 M), and urea (0.04–0.07 M) and 1 μL of the mixed solution was immediately injected into a reaction chamber. We used pyranine as a fluoroprobe to observe the increase in pH due to the formation of ammonia inside the microreactors. For determination of the apparent pH in the droplets, a calibration curve of pH vs. ratio of fluorescence intensities F458/F405 was used (Fig. S4†). Analysis of images was performed with a combination of ImageJ and MATLAB (version R2020a).
Materials
Enzyme labeling and concentration determination:
- AnaTag™ HiLyte™ Fluor 488 Microscale Protein Labeling Kit (Fluorophore labeling kit)
- Phosphate assay kit (Sigma-Aldrich, Cat No: MAK308)
Microfluidic reservoir solutions:
- Inner solution (IS): The inner solution (IS) reservoir contained sucrose (0.2 M), 8-hydroxypyrene-1,3,6-trisulfonic acid (pyranine, 50 μM), urease (50 U mL−1), acetic acid (1 mM), and Pluronic F-127 (0.28%), unless otherwise stated. Calcium chloride (0.1 M and 0.15 M) was also added to the IS for the experiments involving mineral precipitation.
- Middle flow (MF): The lipid-oil solution (middle flow reservoir in the microchannel, MF) contained 6.5 mM 1-Palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine (POPC) dissolved by sonication in mineral oil (MO) and 2% Span-80 for three hours at room temperature using an ultrasonic bath (FB15051, Fisherbrand).
- Outer solution (OS): The outer solution (OS) reservoir contained glucose (0.2 M), glycerol (20%), Pluronic F-127 (0.28%), and acetic acid (1 mM).
- External solution (ES): Acetic acid (1 or 2 mM), glucose (0.2 M), urea (0.04 – 0.07 M)
Equipment
- Ultrasonic bath (Fisherbrand, Cat No: FB15051)
- Pressure-driven pump (Elveflow, OB1 MK3+, Paris, France)
- Microfluidic device (PDMS) (built in-house using soft lithography)
- Confocal microscope (Leica TCS SP8)
- Inverted fluorescence microscope (Etaluma LS560)
- Raman confocal microscope (Bruker, Senterra II)
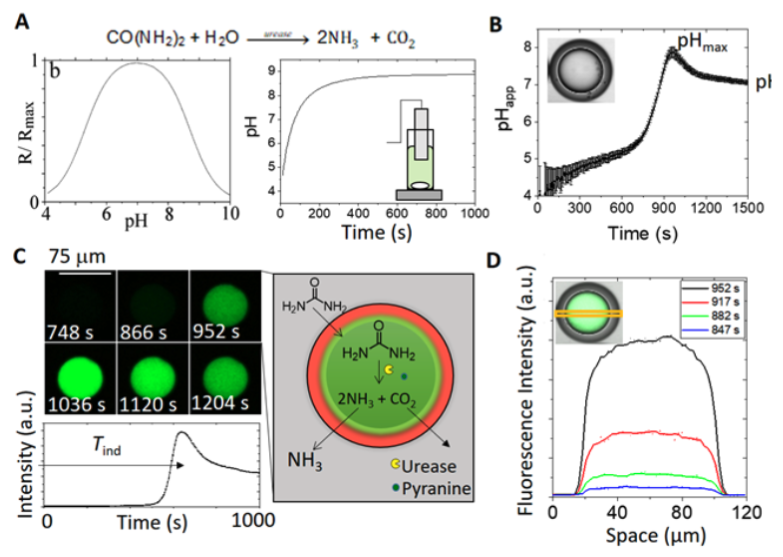
Key Findings | A pH pulse in urease-encapsulated W/O/W double emulsions
The urease reaction has been well studied in batch reactors, and the rate depends on the initial concentration of urea, enzyme, and acid [5]. With relatively high enzyme concentrations, the pH increases rapidly to pH 7 and then more slowly to pH ∼ 9; the enzyme has a bell-shaped rate–pH curve with a maximum at pH 7 (Fig. 3A and S6†). In the double emulsion droplets, Rana and co-workers obtained a distinct pH pulse (not observed in bulk systems) characterized by a lag time or induction time Tind before a sudden increase to the maximum pH (pHmax ∼ 8) and then a slow decrease to the steady-state pH, pHss ∼ 7 (Fig. 3B).
Control of pH-time profile in the W/O/W double emulsions
The characteristic features of the pH time profile, Tind and pHss, can be controlled by manipulation of the transport rates of urea and ammonia between the double emulsion droplet and external solution. In contrast to earlier work with liposomes or polymerosomes, each microreactor behaved virtually independently, and a distribution of induction times was obtained [6].

Here, we used the emulsion properties to control the pH-time profile in the droplets. The pH change rate in the droplets depended on factors including oil shell thickness relative to core size. In general, an increase in the pressure ratio PMF/PIS increases S/C, with smaller core and thicker shells. The clock time increased with increasing shell/core (Fig. 5A, B), and the maximum rate of change of pH decreased (Fig. 5C) due to the longer time taken for urea to cross the oil layer. Unlike in bulk solutions, the reaction can approach a steady state in pH with values below or above 7. The pHss increased with increasing S/C as the ammonia transport out of the droplet was reduced in emulsions with thicker shells (Fig. 5D).
![Figure 5: Control of the pH-time profile with different shell/core (S/C) ratio in the urease µ-reactors double emulsion in the reaction chamber of depth 1 mm. (A) droplets produced with different S/C (0.15 and 0.27) and corresponding average induction times. (B) Apparent pH in time in four droplets with S/C = 0.27. (C) Average rate of rate of pH and (D) Steady-state pH as a function of S/C. Plots show average and standard deviation from 20 droplets. The concentrations were [urea] = 0.07 M, [pyranine] = 50 µM, [AA] = 2 mM, and [urease] = 50 U/ mL, [phosphate]T = 80 mM.](https://elveflow.com/wp-content/uploads/2024/03/Control-of-the-pH-time-profile-with-different-shell_core-_S_C_-ratio-in-the-urease-µ-reactors-double.webp)
Double emulsion platform for pH-regulated production of minerals
The urease reaction is involved in biomineralization, such as the formation of calcite and struvite in bacterial colonies. It has been exploited for environmentally benign production of minerals in various applications [7-10]. Inorganic phosphates have wide-ranging uses in the medical and food industries, including in bone and dental implants, and as drug delivery carriers [11, 12]. The urease-loaded water-oil-water (W/O/W) double emulsion may be used to regulate phosphate precipitation in the droplets, with control of the internal pH achieved by changes in droplet properties.
In batch, stirred solutions with urease in sodium phosphate and calcium chloride, a mixture of precipitates was obtained (Fig. 6A); amorphous calcium phosphate or weakly crystalline hydroxyapatite particles of size ~100 nm and micrometer-sized calcite dumbells. In the double emulsions, we coencapsulated urease with sodium phosphate and calcium chloride. We mixed the droplets with the external solution of urea in acid as described in the previous section. There was an induction period before the rapid appearance of precipitates in the droplets and the crystal polymorph depended on the shell-to-core ratio (Fig. 6B). In droplets with thick shells, spherical hydroxyapatite particles of ~ 500 nm formed, whereas with thinner shells, typically brushite microplatelets of ~ 50 µm.

Conclusion | A new microfluidics-based double emulsion platform for pH control and fine-tuning materials synthesis
In this work, we took advantage of pressure-driven droplet microfluidics (Elvesys OB1) and developed a system of enzyme-encapsulated double emulsion (W/O/W) droplets to obtain a localised pH pulse, with a controllable induction time and steady state in pH. Urease-encapsulated double emulsion droplets of ~ 100 – 200 µm with a mineral oil shell thickness of 10 – 40 µm were prepared using a flow focussing device and the reaction was initiated off-chip by addition of droplets to a solution of urea. The pH increased uniformly and rapidly across the droplets after a time lag controlled by the diffusion of urea and then decreased to a steady state value either above or below pH 7, regulated by the rate of transport of ammonia out of the droplet. A distribution of induction times was obtained in the droplets, thus demonstrating their potential to act as individual (µ)micro-reactors. In contrast, in other encapsulated urease systems, the fast diffusion of ammonia increased the surrounding solution pH [6].
We demonstrated that this approach has potential applications in regulating pH changes for pH-programmable material synthesis in confined environments. We validated that compartmentalization of the urease reaction may be exploited for selecting polymorphs through internal pH regulation. In conclusion, compartmentalization of the urea-urease reaction in double emulsions generates new potentials in developing biocompatible feedback for pH-triggered processes and bioinspired applications in materials science. The microfluidic-based double emulsion platform could be used for fine-tuning material properties, such as pH-triggered organic polymer-phosphate particles for medical applications [13].
References
- Mcdonald JC, Duffy DC, Anderson JR, Chiu DT. Fabrication of microfluidic systems in PDMS. Electrophoresis. 2000;21(1):27-40. https://pubmed.ncbi.nlm.nih.gov/10634468/.
- Xia Y, Whitesides GM. Soft lithography. Angew Chemie – Int Ed. 1998;37(5):550-575. doi:10.1002/(sici)1521-3773(19980316)37:5<550::aid-anie550>3.3.co;2-7
- Trantidou T, Elani Y, Parsons E, Ces O. Hydrophilic surface modification of pdms for droplet microfluidics using a simple, quick, and robust method via pva deposition. Microsystems Nanoeng. 2017;3(April 2016). doi:10.1038/micronano.2016.91
- Wu D, Luo Y, Zhou X, Dai Z, Lin B. Multilayer poly(vinyl acohol)-adsorbed coating on poly(dimethylsiloxane) microfluidic chips for biopolymer separation. Electrophoresis. 2005;26(1):211-218. doi:10.1002/elps.200406157
- Hu G, Pojman JA, Scott SK, Wrobel MM, Taylor AF. Base-Catalyzed Feedback in the Urea−Urease Reaction. J Phys Chem B. 2010;114(44):14059-14063. doi:10.1021/jp106532d
- Miele Y, Jones SJ, Rossi F, Beales PA, Taylor AF. Collective Behavior of Urease pH Clocks in Nano- and Microvesicles Controlled by Fast Ammonia Transport. J Phys Chem Lett. 2022;13(8):1979-1984. doi:10.1021/acs.jpclett.2c00069
- Krajewska B. Urease-aided calcium carbonate mineralization for engineering applications: A review. J Adv Res. 2018;13:59-67. doi:10.1016/j.jare.2017.10.009
- Stocks-Fischer S, Galinat JK, Bang SS. Microbiological precipitation of CaCO3. Soil Biol Biochem. 1999;31(11):1563-1571. doi:10.1016/S0038-0717(99)00082-6
- Barkay T, Schaefer J. Metal and radionuclide bioremediation: issues, considerations and potentials. Curr Opin Microbiol. 2001;4(3):318-323. doi:10.1016/S1369-5274(00)00210-1
- Dhami NK, Reddy MS, Mukherjee MS. Biomineralization of calcium carbonates and their engineered applications: A review. Front Microbiol. 2013;4(OCT):314. doi:10.3389/fmicb.2013.00314
- Habraken W, Habibovic P, Epple M, Bohner M. Calcium phosphates in biomedical applications: materials for the future? Mater Today. 2016;19(2):69-87. doi:https://doi.org/10.1016/j.mattod.2015.10.008
- Sokolova V, Epple M. Biological and Medical Applications of Calcium Phosphate Nanoparticles. Chem – A Eur J. 2021;27(27):7471-7488. doi:https://doi.org/10.1002/chem.202005257
- Cho YS, Moon M, Holló G, Lagzi I, Yang SH. Bioinspired Control of Calcium Phosphate Liesegang Patterns Using Anionic Polyelectrolytes. Langmuir. 2022;38(8):2515-2524. doi:10.1021/acs.langmuir.1c02980

 Job
Job Collaborations
Collaborations Customer
Customer Other
Other
