Heart-on-chip for personalized medicine: CISTEM
Duchenne muscular dystrophy is still poorly understood, and no efficient treatment is available. The promising heart-on-chip models might be a first step towards better managing Duchenne disease.
This project is completed now. If interested, feel free to contact us
Heart-on-chip for personalized medicine: introduction
Duchenne muscular dystrophy occurs in 1/3500 children and leads in 60% of patients to the development of cardiomyopathy, a heart dysfunction, in the second decade of life.
This disease is still poorly understood, and no efficient treatment is available. The use of heart-on-chip models is promising for better management of Duchenne disease.
Organ-on-a-chip, which can mimic the cell microenvironment in a real organ, are animal-free promising systems for a deeper study of this rare disease and for screening potential new drugs.
However, the mechanisms of cardiomyopathy vary from one patient to another, demanding the development of personalized treatments adapted to each patient.
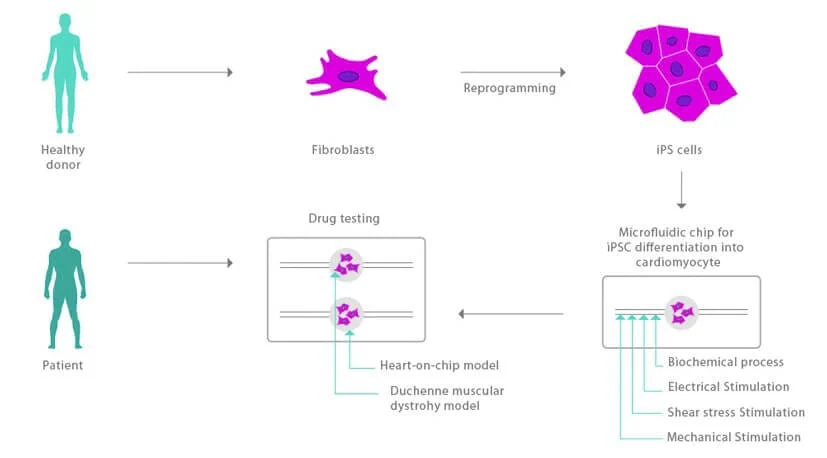
In the CISTEM project, we aim to develop a more precise and personalized heart-on-a-chip model; human-induced pluripotent stem cells are derived in a patient-matched manner in a microfluidic device.
This system will allow precise control of shear stress and electrical stimulation, mechanical strain, and surface morphology.
Heart-on-chip for personalized medicine: project description
We will design and fabricate this microfluidic chip to differentiatepluripotent stem cells at a microscale.
Elveflow pressure controller will handle fluids into this new heart-on-a-chip. Its fast and stable pressure control will allow the precise monitoring of the shear stress applied to the cells, ensuring the creation of biological conditions as closely as possible.
In addition, the possibility to easily configure complex functions for the pressure profile enables the reproduction of the physiological effect of a beating heart. This heart-on-a-chip will allow a better understanding of cardiomyopathy and personalized drug testing.
This project has received funding from the European Union’s Horizon 2020 research and innovation programme under the Marie Sklodowska-Curie grant agreement No 778354 (CISTEM project).


